 
More common with the sponge in both trials, although the frequency of discontinuationįor discomfort differed in the two studies. Rates at 12 months were higher with the sponge than with the diaphragm. trial,Īnd 24.5 for the sponge and 10.9 for the diaphragm in the UK trial. The 12-month cumulative life table termination rates per 100 women for overall In the United States 5 and one in the United Kingdom. Was significantly less effective in preventing overall pregnancy than was theĭiaphragm in the two trials that met analysts’ inclusion criteria, one performed In a recent review of available data, the sponge statistically 4Ĭontraceptive options, the sponge falls below the diaphragm in contraceptiveĮffectiveness. Hours without the insertion of additional spermicide, and it does not requireįitting or a prescription from a health care provider. The diaphragm, the sponge can be used for more than one coital act within 24 To sunburn - are advised to contact a physician immediately.Īre that it is simple to use and noninvasive, and it can be used intermittently Vomiting, diarrhea, muscular pain, dizziness, or rash similar 
Of the warning signs or symptoms of toxic shock syndrome - including fever, Or by women who have ever been diagnosed with toxic shock syndrome. Immediately after childbirth, miscarriage, or other termination of pregnancy It should not be used during menstruation Period after the last act of intercourse). According to theĬompany’s product information, the Today sponge should not be left in placeįor more than 30 hours after insertion (which includes the six-hour waiting Up to 24 hours, no matter how many times intercourse occurs. It is moistened with tap water prior to use and insertedĭeep into the vagina removal is achieved by pulling the attached loop. Made of polyurethane, it contains 1,000 mg of the spermicide 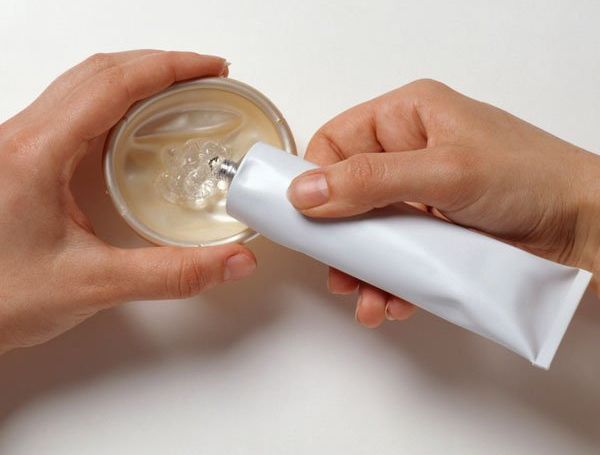
In shape, two inches in diameter, and three-quarters of an inch thick, withĪn attached loop. The FDA also may elect to perform a preapproval inspectionįor manufacturing compliance at the OSG Norwich site, which has an "excellent" Is to change the manufacturing plant to the OSG Norwich site.Ĭompany has product stability material ready for the FDA’s review, which is Which has been submitted to the FDA for its review, says Staab. (NDA) for the sponge remains active however, Allendale Pharmaceuticals must Required to move the company’s request forward? Still is under review, confirms Susan Cruzan, FDA spokeswoman. In repeated delays and a switch from a contract manufacturer in Mainland, PA, Tougher new FDA standards for manufacturing and record keeping resulted Originally planned to reintroduce the sponge in the American market in fall Allendale Pharmaceuticals acquired manufacturing and marketing rights Problems caused by water quality issues at the old factory where the sponge Of New York City, ceased production when it determined it cost too much to correct The sponge from the market its former manufacturer, Whitehall-Robins Healthcare Of the sponge’s re-emergence some 250 million sponges were sold between Today’s While the sponge is not for sale in the United States,Īmerican women are ordering the device from the Canadian web sites. equivalent of about $2.90Įach on the two web sites. He predicted it would be on Canadian pharmacy shelves and other mass-merchandising At Contraceptive Technology Update press time, see vaginal contraceptive?įDA approval for marketing in United StatesĪnnouncement to revive the Today contraceptive sponge, the company responsibleįor its rebirth finally has released the product in Canada and says it is pressingįor Food and Drug (FDA) approval to market the device over the counter in theįor sale over the Internet on two Canadian web sites, and says Robert Staab, PhD, chairman and chief scientific officer of Allendale 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
